A New Rx for Pharma Cold Chains

Pharmaceutical supply chains are adjusting to meet the demands of fast-changing biologics medicine.
Ensuring product integrity and security throughout the supply chain has always been a high priority for pharmaceutical manufacturers. In the past few years, however, executing this mission has grown far more challenging. New market dynamics—including a changing product portfolio, stricter regulations, geographically extended supply chains, increasing risk, and intense cost pressure—have significantly raised the stakes.
The challenge at the heart of this new business paradigm is the fact that drug-makers today face an exponential growth in the need for temperature-assured distribution and handling of materials, from active ingredients to finished products. This requirement stems from a dramatic change in the nature of pharmaceutical and biotechnology products.
Specifically, drug portfolios are evolving away from reliance on small molecule/chemical pharmaceuticals and toward more structurally complex biotechnology drugs, which have much stricter temperature requirements. In addition, new regulations in the United States, the European Union, and elsewhere, have dramatically expanded the list of drugs that require temperature-controlled handling—in particular, stipulating new rules for handling products that fall into the controlled ambient (15 to 25C) category.
The stakes in this new pharma world order are high. “Gaps and breakdowns in good distribution practices can trigger a chain of dire consequences for manufacturers—from increased regulatory scrutiny and steep financial penalties to slumping sales, a surge in shareholder apprehension, an irreversibly damaged brand and reputation, and worst of all, compromised patient safety,” say Jamie T. Hintlian and Ryan Kelly of consulting firm EY in their report A Roadmap for Risky Territory.
Additionally, the growing practice of healthcare providers and payers (insurance companies and governments) basing purchasing and reimbursement decisions on a drug’s therapeutic performance means it is critical that manufacturers protect product efficacy throughout the supply chain.
In the face of these realities, manufacturers have gone back to the drawing board to re-think their temperature-controlled supply chains.
THE BIOLOGICS BOOM
While the growth in drug spending worldwide is healthy in aggregate, the rise in spending on biologics and specialty drugs is far more dramatic. In the United States, for example, expenditures for these new specialty drugs are expected to significantly increase between now and 2020 (see Figure 1). This same trend is playing out across the world, as the transition toward more structurally complex and temperature-sensitive drugs gathers momentum.
The industry’s migration to these new medicines injects tremendous complexity into the supply chain. For example:
- Product must be handled within specific condition tolerances—i.e., cold chain, controlled room temperature (CRT), and frozen (see Figure 2). Failure to maintain appropriate conditions at any point in the supply chain can harm the efficacy of the drug, result in the loss of a shipment, and put patients at risk.
- Biotechnology medicines often are extremely high value. Annual per-patient treatment costs can exceed $100,000. This means a single consignment may be worth upwards of $50 million.
- Because drugs are manufactured in specialized locations, these temperature-controlled products frequently traverse the world on their way to market. They move through a sometimes extreme range of climactic zones while en route, and travel via multiple modes with numerous hand-offs.
“It all boils down to one simple fact,” says Jay McHarg, president of American Aerogel, a manufacturer of temperature-controlled packaging solutions based in Rochester, N.Y. “There are far more temperature-sensitive products on the market today than there were even three years ago. In biopharma, 80 percent of the top products introduced in the past two or three years are temperature sensitive.”
Global distribution of these perishable products increases risk. And in emerging markets, infrastructure issues—lack of proper temperature-controlled facilities, transport options, and handling capabilities, as well as higher ambient and container temperatures—come into play.
“All of this means product protection—preventing damage and/or spoilage—is high on the pharmaceutical company agenda,” reports Jonathan Blamey, vice president, Global Solution Design, DHL Life Sciences & Healthcare.
Not surprisingly, expenditures for cold chain logistics are climbing. The total size of the healthcare cold chain logistics services market is expected to grow from $8.5 billionto nearly$13.4 billionby 2020, according to IMARC Group’s Global Healthcare Cold Chain Logistics Market Report & Forecast (2016-2020).
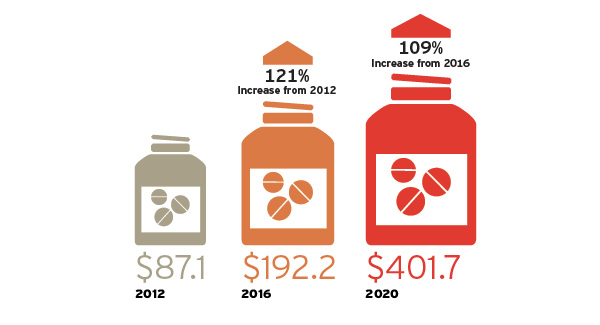
Figure 1: Projected Specialty Drug Spending 2012 to 2020
Spending amounts in U.S. billions
U.S. expenditures for new specialty drugs are expected to substantially increase by 2020. A similar trend is playing out across the world.
Source: PwC Health Research Institute estimates based on data from CVS Caremark, 2014
THE COMPLIANCE SQUEEZE
As the nature of pharmaceutical products is changing, so are the global regulatory regimes that control them. Pressure is increasing to ensure “ship-to-label” regulations are met. Authorities require proof that products have not only been stored at the temperature stated on the label, but also kept within an approved temperature range during transportation.
This stepped-up regulation is largely the result of revised guidelines the European Commission issued in 2013, which established good distribution practices (GDP) requirements for pharmaceutical products. These rules are fast becoming the de-facto standard around the world.
In addition to extending enforcement to include transportation as well as storage, the European Union GDP rules greatly expanded compliance oversight to include medicines not previously covered by temperature-control regulations. The GDP rules do not specify exact procedures. Instead, they focus on a risk-based approach to managing condition outcomes in adherence to label requirements. This makes compliance a challenge.
“There’s a lot of interpretation about what to do and how the enforcement will affect us,” says one European pharmaceutical manufacturer. “Authorities are not telling pharma companies anything until they do an inspection; they have to figure out the answers.”
Expanding compliance requirements, particularly to CRT products, is straining supply chain operations as well as budgets. “Right now, everyone’s costs are going up,” reports the director of specialty and cold chain logistics at one major U.S. drug-maker. This fuels the urgency of developing a more cost-effective temperature-controlled life sciences supply chain.
As companies adapt to this new product and regulatory environment, temperature-controlled supply chains must incorporate ways of mitigating risk and loss, have strong contingency capability, and deliver proactive problem-resolution processes. And they must be segmented based on tiered product value, handling needs, customer service requirements, and compliance rules.
Beyond this, emerging best-in-class life sciences cold chains are adopting the following four key strategies.
- A specialized and compliant network. The unique requirements of cold chain pharmaceuticals demand a highly specialized and compliant network tuned to moving product efficiently, while protecting its integrity. This network consists of the facilities and assets required to handle temperature-controlled cold chain pharmaceuticals, as well as the IT systems needed to monitor and manage global product flows.An intelligent IT platform underpins the physical network. Monitoring solutions establish visibility checkpoints that enable more proactive control of the shipment, and allow for intervention should an adverse situation arise. Because the IT platform houses the manufacturer’s standard operating procedures (SOPs), these interventions conform to regulatory and company requirements.
On a strategic level, intelligent IT networks harness the power of big data and analytics to reduce risk and make better decisions about managing the temperature-controlled supply chain. “Because we have collected, aggregated, and analyzed data from multiple customers, thousands of shipments, hundreds of trade lanes, and numerous types of packaging, we can identify risk trends and design the supply chain process to prevent, avoid, or mitigate those risks,” explains David Bang, global head, DHL Temperature Management Solutions.
Assets and IT provide the physical network to manage goods, but the people, and their expert knowledge of handling pharmaceutical products, make the network work. “First and foremost,” says the U.S. pharma company’s cold chain director, “we expect people to understand that what they are handling is not just a carton of nuts and bolts. A life may depend on it.”
This means training is a top priority. “We have trained more than 3,000 employees on GDP as part of our ongoing program,” reports Nigel Wing, vice president, global head, DHL Life Sciences & Healthcare. “It’s not enough for us to have a best-in-class cold chain infrastructure; we must constantly invest in the people that work within it.”
- Globally consistent processes. Effective temperature-controlled supply chains rely on well-defined SOPs to make them work. The foundation for these SOPs is comprehensive supply chain risk assessment. SOPs address the product characteristics, season, weather conditions, in-transit and in-storage condition requirements, documentation needs, in-transit monitoring, and compliance.”You need to understand your risks before you put product into the supply chain,” explains Perry MacDonald, warehouse and export manager for Pfizer Global Supply Hospira. “Conduct risk assessments collaboratively with your partners, and build policies and procedures around those risk findings. These include mitigating actions—what happens to my product if the aircraft is delayed or breaks down? They also include lessons learned from past incidents.”
MacDonald asks his third-party logistics (3PL) providers to develop their own risk assessment of his products and lanes. He and the 3PL then blend that assessment with Hospira’s procedures to arrive at mutually agreed-upon SOPs.
“This collaborative approach is important, especially if you’re making a change in your supply chain,” MacDonald insists. “The greater the risk in the supply chain, the tighter the collaboration on SOPs between the manufacturer and the forwarder needs to be.”
Kevin Tuel, senior manager of global sourcing at CSL Behring wholeheartedly agrees with MacDonald’s assessment. CSL Behring manufactures plasma protein bio therapeutics, which are used to treat conditions such as hemophilia, immune deficiencies, sepsis, and shock. Every one of its products is temperature controlled (frozen), moving around the world between production and plasma collection facilities in Switzerland, Germany, the United States, and Australia.
Tuel worked closely with the company’s global freight forwarder to develop SOPs for every shipping lane in this complex supply chain. As he explains, these lane SOPs must be filed with and approved by the regulatory authorities in Europe and the United States.
“This is a complex process,” he acknowledges. “We had to agree on every detail. At first, we wanted to control the trucking portions of the shipment lanes ourselves, but because of liability and risk issues, we finally decided to let our forwarder provide the complete door-to-door solution and control the moves end-to-end.”
For global pharma companies, SOPs are a constant work in progress. “We are always fine-tuning our SOPs to optimize and improve the way we handle our flows,” notes Aurelien Sarazin, distribution manager-EMEA APAC, for Bristol-Myers Squibb Co.
- Risk-appropriate packaging. The third essential of a smarter temperature-controlled supply chain is packaging. In principle, shippers of temperature-controlled pharmaceuticals have a broad choice of options. Essentially, packaging falls into two basic categories: active and passive.Active systems range from discrete packages and full containers/trailers to entire aircraft. They use an energy source combined with thermostatic control to maintain temperature. Passive packaging solutions look like conventional packages but use materials such as water/ice or dry ice to keep products at the desired temperature. Unlike active solutions, passive packaging does not respond actively to adverse temperature conditions.
The choice of packaging is a matter of balancing cost with the risks and benefits of a particular option. When making their selection, manufacturers must consider the value of the pharmaceutical product, its temperature-management needs, regulatory compliance requirements, customer and market risk, and total cost. “The analysis is based on a single question: How much risk are you willing to assume?” says Joachim Kuhn, CEO of thermal packaging company va-Q-tec.
Fortunately for the drug companies, stricter temperature control regulations have sparked a new wave of innovation in packaging technology. “Six or seven years ago, choices were limited,” says McHarg. “Only two or three companies made temperature sensing/monitoring devices, for example. They were expensive and cumbersome.”
Today, however, new technologies can monitor temperature and report back a real-time stream of condition information—all at a far more affordable price point.
CSL Behring sees the benefits of these new technologies firsthand. “We had been using temperature monitors in our containers,” Tuel recalls, “but would find out about a temperature excursion only after the container arrived, was opened, and temperature data was downloaded. That’s too late.
“We need to see a problem while the shipment is in transit, so we can do something to save it,” he adds. “A pallet load of our product can be worth more than $1 million, so we can’t afford to lose it. Losing a clinical trial shipment due to a temperature excursion can set back the launch date of a new drug by months. That’s costly.”
To tackle this issue, Tuel recently started working with Verizon on remote monitoring of CSL Behring’s ocean containers in transit—with good results.
These new technologies, and the regulations that spawned them, have helped the entire pharma supply chain get better at protecting product integrity. But while packaging—even the most expensive active solutions—protects the product, it isn’t the complete answer. “Even the most robust packaging material is no good if the handling is poor,” says Wing of DHL. “It takes people to make the cold chain work, so it comes back to having good SOPs, knowledge about the product, and constant training.”
- Total cost strategy. Smarter cost management is the fourth essential of the next-generation life sciences cold chain. “The whole industry—our customers and all partners along the supply chain—are on a journey together to drive down total costs,” Wing says. Leading manufacturers and their logistics partners are evolving away from simply managing costs on a purchase-price basis, to a total cost of ownership (TCO) model.
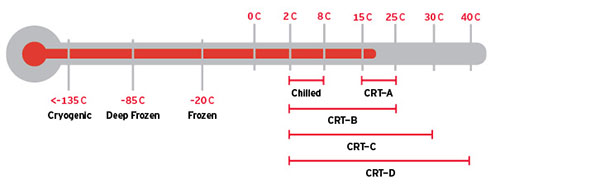
Figure 2: Common Product Temperature Ranges Within a Controlled Supply Chain
Maintaining recommended temperatures throughout the supply chain ensures the efficacy of drugs.
CRT: Controlled Room Temperature
Source: DHL Global Forwarding, 2015
Strictly defined, TCO is the compilation of direct and indirect costs associated with a purchase, transaction, or activity. In the case of the temperature-controlled pharmaceuticals supply chain, TCO carries a much broader definition to include everything from patient safety to product and market share losses to brand risk.
One way of thinking about TCO is to view it as an iceberg (see Figure 3). Direct costs, such as the invoice value of a service, represent only a fraction of the whole cost picture. The hidden costs—patient safety, product loss, new product launch delays—carry the real risk to the business.
Identifying and calculating these hidden costs, and factoring them into a business decision or operating plan, is not easy. “Within large pharma organizations, costs often are spread across various departments, further hindering a strategic approach to cost management,” says Angelos Orfanos, president, DHL Life Sciences & Healthcare. “A damaged or lost product is one cost line, the cost of packaging is in a different line, transportation is in yet another line, and so on.”
Economizing on one aspect of the temperature-controlled supply chain without factoring in the total cost can and often does backfire. Valgeir Pétursson, managing director of Malta Principle Trading Company/Pharmapack & International Logistics at Allergan, offers an example. “If a logistics manager sends a shipment of five pallets on a dedicated truck, let’s say that costs approximately $4,300. If he puts that same product on a consolidated shipment, the freight rate is far lower—approximately $540 to $1,000. The manager is very happy with his savings—until the consolidated load is ruined by a temperature excursion. He has now lost about $218,000 worth of product by trying to save $3,300.”
The tangible loss is almost nominal when compared to the total cost of the loss. “If you’re not able to provide product, someone else will, so you lose that sales opportunity,” Pétursson says. “More importantly, once customers start using another product, many won’t change, so you’ve lost that market share permanently. That can translate into millions in lost revenue.”
Figure 3: Elements of the Total Cost Approach Iceberg
Source: DHL 2015
PARTNERING FOR SUCCESS
Taken together, the four essentials of the smarter life sciences supply chain drive powerful benefits. They safeguard product, effectively manage complexity and risk, reduce total cost, and improve profitability and competitiveness.
“A consistent global approach to managing the new temperature-controlled pharma supply chain is the most effective,” McHarg believes. “Many large pharma companies are doing just that—mapping all of their shipment lanes, grouping them into channel types, and specifying approved solutions for each channel type.Supply chain decisions then become more standardized and manageable.”
Beyond executing the specifics of infrastructure, data analytics, people, packaging, and total cost management, the success of a temperature-controlled pharmaceutical supply chain comes down to one powerful concept: collaboration.
“Our most successful supply chain outcomes come from when we have good collaboration along the whole chain,” says Mattias Almgren, deputy CEO of container manufacturer Envirotainer. “The more we are able to break down the walls between shippers, airlines, freight forwarders, packaging suppliers—the more we all succeed.”
Editor’s note: This article is based, in part, on a recent whitepaper—The Smarter Cold Chain: Four Essentials Every Company Should Adopt, written by Lisa Harrington and published by DHL.