The Race is On: Chain of Custody in the Pharmaceutical Supply Chain

New regulations ensuring the integrity of the pharma supply chain are nearing the finish line. While some companies have taken an early lead to compliance, others are late getting out of the gate. Which team are you on?
Top 10 Health Supply Chain Issues
The Last Word on Chain of Custody
The Healthcare Supply Chain: Are You Connected?
DSCSA Timeline for Manufacturers
Tracking the global path of a pharmaceutical product from raw material to end consumer is complicated and risky. When done right, strategic chain of custody solutions can leverage brand reputation. But persistent vulnerabilities—poor visibility, and tainted, expired, or counterfeit drugs—can lead to numerous outcomes far more costly than a damaged reputation. Bad deliverables can be the difference between life and death.
Major pharmaceutical companies are susceptible to grim setbacks when weak points infiltrate the chain of custody. Some of these setbacks have become highly publicized cases that resulted in federal criminal charges.
One infamous example occurred in March 2015, when McNeil-PPC Inc., a wholly owned subsidiary of Johnson & Johnson, pled guilty in Federal District Court in connection with adulterated infants’ and children’s over-the-counter (OTC) liquid drugs. The ruling came five years after the Tylenol maker announced mass recalls of OTC medications including infants’ and children’s Tylenol and infants’ and children’s Motrin. In a 2010 press release, McNeil revealed that some of the unexpired recalled medications might have contained "tiny particles." Those foreign specks were later identified as metal—nickel/chromium-rich inclusions that were not intended ingredients of the medications.
In a statement issued after McNeil’s guilty plea, FDA Commissioner Margaret A. Hamburg, M.D., said, "The Food and Drug Administration expects manufacturers to have systems in place that will quickly discover and correct problems in medical products before they enter the U.S. marketplace. Today’s guilty plea holds accountable those corporations who risk jeopardizing public health by not adhering to the high standards set by drug manufacturers."
Another threat to drug businesses lacking chain of custody systems include counterfeit medications, which have resulted in some deadly and sinister consequences. In 2006, the U.S. Attorney’s Office indicted 18 people for a "multi-million-dollar international conspiracy to smuggle untaxed cigarettes, counterfeit Viagra, and other goods to raise money for the Middle East terrorist group Hezbollah," according to a brochure published by Viagra maker Pfizer, which has fallen victim to multiple counterfeit products, including Lipitor, Celebrex, and Zoloft.
Pfizer’s brochure also outlines the various ways that counterfeits can infiltrate business, and describes how drugs may move sideways from authorized distributors to middlemen or secondary wholesalers who sell drugs to one another, even within the United States. As part of its chain of custody process, Pfizer uses radio frequency identification (RFID) tags to identify and track its highly counterfeited products.
Amid these ongoing challenges, recent federal legislation is transforming all aspects of the pharma industry concerning track-and-trace solutions. The FDA’s Drug Supply Chain Security Act (DSCSA), Title II of the Drug Quality and Security Act signed into law in late 2013, outlines imperative steps to building "an electronic, interoperable system to identify and trace certain specific drugs as they are distributed in the United States."
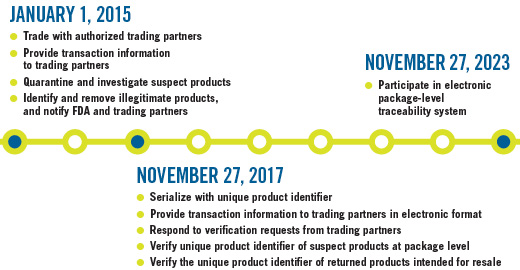
The multi-tiered act, which will be enacted in phases through 2023 (see timeline, page 70), requires all supply chain sectors—including manufacturers, re-packagers, wholesale distributors, and dispensers—to take part. While the act instills universal compliance, it also poses new challenges to businesses that will need to add improved, additional layers of management to their supply chains—inciting a race against time as the act rolls out.
"The DSCSA is topical, but it represents a need to fully secure the supply chain globally," says Robin Hooker, director of healthcare sector marketing at UPS. "We’ve seen the need for more supply chains to be global and to offer end-to-end solutions." As the population continues to expand due to a growing middle class, the need for better synchronization across global marketplaces is also rising steadily, Hooker adds.
Another DSCSA mandate, set to take effect in 2017, requires adherence to serialization measures, in which manufacturers must place unique product identifiers on drug packages to verify legitimacy. According to the law, wholesalers will not be able to accept products that are not serialized. "Serialization is on the horizon, and will come more quickly than businesses are prepared for," Hooker notes.
While serialization will start playing more of a role in unifying the line of communication across the supply chain, it’s not the means to all ends.
"It has been frustrating to see all the attention—and budgets—being devoted to serialization, along with the dawning realization that without authentication, serialization is an inadequate fix for tracking products," explains Dr. Sharon Flank, CEO of InfraTrac, a Silver Spring, Md.-based company that provides light-based verification solutions to protect brands against counterfeiting.
"The first legal line of defense in the United States is serialization," Dr. Flank says. "The validation infrastructure is complex and expensive, but without it, the number codes have no data security and don’t protect much.
"The need for authentication is growing as well," she adds. "It puts fast, one-step checking into the hands of multiple stakeholders, including distributors, border agents, hospitals, and re-packagers."
Prepping for Change
Managing a balanced chain of custody is integral to adhering to all elements of the DSCSA as other requirements take effect within the next decade. Key strategies—and potential missteps—can either help synchronize or disrupt the flow of all parties involved in the chain of custody process.
Good companies sometimes make bad decisions when attempting to expedite steps within the chain of custody. "Every business faces a time when it needs to run with a snap decision, but complex processes such as chain of custody do not fall into that realm," contends Tom Wengrowski, executive vice president at TRIOSE, a Wyomissing, Pa.-based logistics management firm specializing in managed solutions for the healthcare industry.
From his experience working with healthcare companies, Wengrowski cites two detriments to the chain of custody process: poor planning horizon and resourcing. "For example, companies know that they will be required to report a piece of information by a certain date in two years," he says. "But instead of developing a plan early, companies procrastinate due to a lack of resources." The result: serious time compression.
Companies must also dedicate time to understanding the project’s definition and requirements. "Even great companies sometimes don’t take the time to apply the right resources to defining the requirements," he explains. "Jumping into a solution is a common mistake."
To avoid this type of error, Wengrowski recommends investing time to clearly understand the requirements or problem, analyze your own capabilities, and examine the technology and the people supporting it. This will help determine if your current process is sufficient or if it needs to change.
Businesses should also seek outside opinions on the solution. "Those who develop complex solutions are often too close to the challenges to be objective," Wengrowski explains.
Visibility within the chain of custody is another prominent challenge. In fact, a recent study, Top Ten Global Health Supply Chain Issues, reveals that shipment visibility was measured as "important" and "critical" by 83 percent of survey respondents" (see sidebar, below).
"Past the central or national level, communication and shipment visibility deteriorate as products move farther down in-country supply chains," according to the report, which also notes one alarming, albeit well-known fact: recipients at most supply chain stages—including health facilities and intermediate warehouses—have no information on when orders will arrive.
"Everybody can get visibility to shipments, but unfortunately, in the pharmaceutical world, visibility might also mean that your product could be destroyed," says Phil Abbate, global vice president, pharmaceutical and healthcare, for global 3PL UTi Worldwide.
Abbate adds that he constantly conveys to his operational staff that visibility alone is no longer adequate. "What we need is visibility with interventional processes," he notes.
"We don’t think of visibility as track and trace, but rather as having visibility into every business object," says Diane Palmquist, vice president of manufacturing industry solutions at GT Nexus, an Oakland, Calif.-based company that offers a cloud-based platform for supply chain management. Companies need to assess costs, service levels, service commitments, and other factors related to the business object, whether that means an order, a shipment, or an invoice, she says.
"To have good visibility, and especially to detect counterfeit drugs in the chain of custody, gathering data from all parties is essential," Palmquist says. "Then you have to integrate and analyze that data, which provides predictive analytics for future use.
"If you see that a product is not traveling on its specified route, you have to be able to take action," she says.
To Air is Human
Visibility also presents a challenge because pharma shipments are subject to extensive network handlers across the globe, leaving room for multiple mistakes. The issue is especially prominent in temperature-controlled pharmaceutical supply airfreight processes, where most temperature exceptions occur.
"Shipping pharmaceuticals via air is complicated because some nationalized airport operators have a closed system that limits visibility to what is happening to a package," explains Jon Edgcombe, software group leader at Boston-based Cambridge Consultants, a product design and development firm that provides cold-chain solutions for the healthcare industry. He adds that the process inevitably involves handovers—where drugs come out of a well-controlled plane environment, then sit on the tarmac, only to be placed into a well-controlled environment for the rest of their journey—and is where end-to-end monitoring comes into its own.
"Monitoring a drug shipment’s journey is of increasing importance as the industry gradually moves to optimized shipping lanes—where substantial cost savings can be achieved while maintaining good temperature performance," Edgcombe explains. "But that’s only if enough is known about the individual lanes across an annual cycle."
Strengthening the Chain
Edgcombe also notes that the current chain of custody system—specifically relevant to a shipment’s last mile—is often subject to a lack of responsibility, authority, and accountability.
"One way to strengthen the chain of custody process is for the organization that ultimately carries the liability to deploy a solid track-and-trace system by using one core identifier that carries through from manufacture to dispense or point of care," he says. The identifier can be associated with GS1 and logistics track-and-trace numbers to tie to existing expiry date and batch code systems with minimal system modification.
Cambridge Consultants has added some space on its logging devices for custom identifiers to be stored locally, paired with the device serial number, and reported back to data collection systems. This helps tie up the records for improved compliance with chain of custody processes, Edgcombe explains.
On the parcel level (which includes moving through a small package network, before packaging occurs) a network profile analysis is one essential element that needs to be set in place, says Hooker.
"UPS has invested heavily in sending data loggers through tracking packages during some of the hottest and coldest periods throughout the year," he says. "We do this on a global basis to understand the environment that our parcels are moving through."
"Cold chain or temperature-sensitive packaging moving through the UPS network carries a certified label that allows some firms that may not have in-house testing capabilities to plug into UPS’ network analysis and validation of some manufacturers’ packaging. That’s critical," he adds.
UPS met with some large firms that were accessing big data repositories based on broad ambient temperatures drawn from weather reports. But those temperatures are markedly different from the actual temperature in the small parcel environment, Hooker explains.
The good news for industry is technology cost efficiency, driven by sweeping changes across the consumer industry. "In particular, the cost for MEMS technologies such as accelerometers—as well as the silicon area required for temperature, relative humidity monitoring, digital signature technology, and short range wireless communications such as Bluetooth Low Energy—have all been coming down, enabling a new generation of sensors that are good at a few key functions, but can benefit from the scale of being used in billions of consumer devices, Edgcombe explains.
Experts say that infrastructure costs are dropping as certain devices (think smartphones) are increasingly used for data capture.
"As computing power increases—even low-cost processors sold in high volume—new solutions become available that use algorithms to process raw sensor data into information and context before sending data wirelessly," says Edgcombe. "This enables a new generation of battery powered Internet of Things devices to provide increased visibility and quality."
Edgcombe also recommends horizon scanning for new technologies in order to ensure tangible benefits can be quickly deployed across the organization.
With more opportunities for technology that bridges the communication line to take center stage Abbate warns companies to proceed with caution. "A company can implement all the technology in the world, but without process controls, they might as well throw it away." His company works with a developing risk-aversive tool for cold chain pharmaceuticals, an operational tool (at the desk level) that would put in the risk factors on any one given move.
UTi is currently working on the development of active temperature management. "As technology develops, active temperature management systems are something that we’ve been looking at for two years," he says.
Aside from visibility and technological strategy, other deep issues can be reversed with better management. One such common error is not allowing enough time for due diligence.
Companies must take the time to vet new solutions and partners. "Many large companies rely on a strict RFP process to vet providers, but a poorly constructed RFP will lead to a poor outcome," says Wengrowski. "I can’t tell you how many times I’ve received an RFP from a company that says it needs a response in two weeks, will make a decision in 30 days, and wants to be up and running in 60 days. This timeframe is unrealistic; complex processes can take 90 to 120 days to implement alone."
Digging Deeper
He also says companies can do more homework when they get background information from prospective technology partners. "Getting a list of current clients is great, but truly understanding what those clients get from that partner, and how big they are, is more important."
Jeff Liter, senior principal in West Monroe Partners’ merger and acquisitions group, has more than 35 years of experience in operations, integration and restructuring, strategy, and profit and loss management. He offers a different perspective on the chain of custody supply chain: "Much like real estate lives by the credo location, location, location; the first step in chain of custody best practices is document, document, document," he says.
Liter also advises companies to build a strong external and internal audit program to monitor compliance, and to develop a traceability matrix for all key raw materials and ensure that it is always up to date.
Final steps include selecting the right individuals to manage chain of custody issues. "Put complete and detailed CAPA procedures in place, where trained and competent people are charged with getting to the root cause analysis of any deviations or nonconformance," he says.
"Finally, education and training are key to ensuring people have a deep understanding of why the extra precautions are in place," Liter adds. "Reinforcement in a manner that invokes effective learning is a must."
Top 10 Health Supply Chain Issues
Recent research published in the academic journal Operations Research for Health Care titled Top Ten Global Health Supply Chain Issues provides insight into the leading challenges across the pharmaceutical supply chain. Through surveys and interviews with 22 industry stakeholders with roles in the global health and pharmaceutical supply chain—ranging from manufacturer to individual facility, the researchers identified key global health pharmaceutical delivery challenges ranked in order of most critical:
1. Lack of coordination
2. Inventory management
3. Absent demand information
4. Human resource dependency
5. Order management
6. Shortage avoidance
7. Expiration
8. Warehouse management
9. Temperature control
10. Shipment visibility
The authors, who restricted the survey to the delivery of pharmaceutical products in developing regions instead of the “broader overall delivery or health services and products throughout the globe,” conclude that they are hopeful that the findings “will drive future actions, policies, and research that can ultimately improve pharmaceutical delivery in developing regions and save lives.”
The Last Word on Chain of Custody
Understanding the terminology around chain of custody issues is critical to compliance. This glossary explains some key terms and definitions.
Downstream: The directional order in which pharmaceutical product flows—and transactions occur—across the supply chain (from manufacturers to repackagers to wholesaler distributors to dispensers).
Homogeneous case: A sealed case containing only product that has a single National Drug Code number belonging to a single lot.
Interoperable electronic system: System to capture and exchange transaction data for each sale.
National Drug Code: A unique 10-digit, three-segment numeric identifier assigned to each medication prepared for commercial distribution in the United States. The three segments of the NDC identify the labeler, the product, and the commercial package size.
Product identifier: A standardized graphic that includes the standardized numerical identifier, lot number, and expiration date of the product.
Standardized numerical identifier: A set of numbers or characters used to uniquely identify a package or homogeneous case of product; includes a National Drug Code combined with a unique alphanumeric serial number of up to 20 characters.
Transaction history: A statement in paper or electronic form that includes the transaction information for each transaction going back to the point of origin by the manufacturer of the product.
Transaction information: All of the information associated with a transaction including: the name or names of the product, the strength and dosage form of the product, the container size, the number of containers, the lot number, the date of the transaction, the date of the shipment, the business name and address from which ownership is being transferred, and the business name and address to which ownership is being transferred.
Transaction statement: A statement, in paper or electronic form, confirming that the trading partner who is transferring ownership is authorized as required by law, received the product from an authorized person or entity as required by law, received transaction information and a transaction statement from the prior owner of the product, did not knowingly ship a suspect or illegitimate product, did not knowingly provide false transaction information, and did not knowingly alter the transaction history.
Trading partner: A manufacturer, repackager, wholesale distributor, or dispenser from whom a manufacturer, repackager, wholesale distributor, or dispenser accepts ownership of a product.
Upstream: The opposite directional order in which pharmaceutical product flows—and transactions occur—across the supply chain (from dispensers to wholesaler distributors to repackagers to manufacturers).
SOURCE: The PEW Charitable Trusts
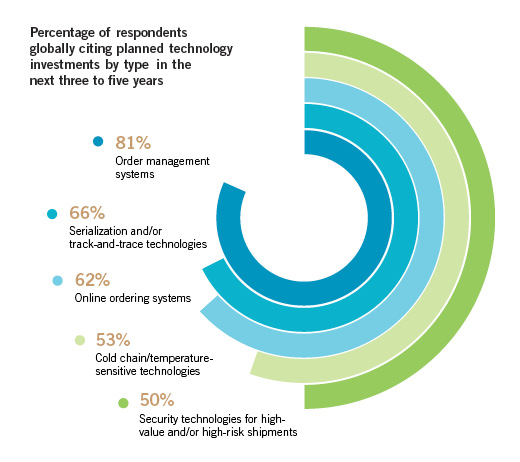
The Healthcare Supply Chain: Are You Connected?
Investing in technology may be a winning strategy in healthcare, according to UPS’s annual Pain in the (Supply) Chain Survey. When survey respondents were asked about specific planned technology investments, two common themes emerged: Front-end technology investments, reflecting the need for manufacturers to pay attention to technology’s role at the front end of their operations; and product protection investments, illustrating a focus on ensuring product integrity and addressing product security concerns.
DSCSA Timeline for Manufacturers
Pharmaceutical manufacturers, the first step in the distribution supply chain, develop and produce prescription drugs for consumers and patients. They ship finished medicines to repackagers, wholesale distributors, and directly to dispensers.
Source: The PEW Charitable Trusts, April 2014